How do you selectively address an individual molecule?
What follows is a brief discussion of the two main factors which lead to the selective detection of a chromophore in the condensed phase by fluorescence excitation spectroscopy: spatial selection and spectral selection.

Spatial selection: The first important step on the way to single-molecule sensitivity is to limit the “active” volume in the sample, i.e. the part of the sample which is illuminated by the excitation light (excitation volume) and from which fluorescence and scattered laser light are collected and guided to the detector (detection volume). This is mainly influenced by the thickness and optical quality of the sample and by the imaging quality of the optics which are used to focus the excitation light and collect the fluorescence. Samples of particularly high optical quality are molecular crystals, frozen n-alkane solutions and spin-coated polymer films, but high quality optics also allow single-molecule investigations of systems with inherently poorer optical quality, such as living cells and fluid solutions of biomolecules with approximately physiological composition. All the techniques described in the introduction to optical microscopy can achieve single-molecule sensitivity; wide-field imaging and confocal microscopy, which are both diffraction limited, as well as near-field microscopy, which achieves a reduction of the illuminated volume beyond that limit.
Spectral selection: Even with the best possible optics we can never even come close to a resolution corresponding to the dimensions of a single molecule (sub-nanometer scale). To be more precise: In a typical apparatus we have to deal with about 109 molecules in the excitation volume when we investigate a condensed-phase sample. Clearly we need a second factor besides spatial isolation in order to reach the single-molecule regime; this is achieved by spectral selection: We have to make sure that the many molecules in the focus cannot interact resonantly with the laser – except for the one we are interested in, of course. Therefore we prepare the chromophores which we want to investigate as a highly diluted sample in a matrix which does not absorb the wavelength at which the chromophores are resonant. (We can easily eliminate almost all of the scattered laser light by using filters in the detection path.) This way we make use of the fact that fluorescence is such a strong and selective interaction (see also the next chapter).
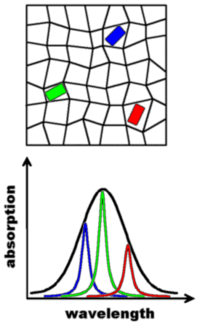
A special case of spectral selection is encountered in low-temperature single-molecule spectroscopy (typically below 10 K): If a suitable chromophore/matrix combination is chosen it is feasible to observe extremely narrow “zero-phonon” lines, the width of which can be as low as 10-5 nm. This is possible since cryogenic conditions can “freeze” the dynamics in the matrix which normally broaden the molecular absorption lines. The static disorder of the host matrix is still present at low temperatures, on the other hand, and therefore every guest molecule experiences a slightly different environment. This is symbolized in the illustration above by the distorted grid in which the three guests are embedded. The different environments lead to different resonance frequencies for the guest molecules because their electronic energy levels are sensitive to their immediate surroundings. The resulting spread in absorption wavelength is called inhomogeneous broadening and it can be several orders of magnitude larger than the width of the absorption lines of the guests. The figure above depicts a sample with a still fairly high concentration of chromophores; if the concentration is reduced the guests can be distinguished by their absorption frequency – even though they are all the same chemical species. This is possible by using narrow-bandwidth laser sources which can be selectively tuned into resonance with a given chromophore. Please keep in mind the difference between the two types of spectral selection discussed here: The distinction between non-fluorescence matrix molecules and the chromophores makes use of differences in absorption wavelength of hundreds of nanometers, while the spectral selection at low temperatures relies on differences in absorption wavelength on the order of 10-4-10-5 nm and is able to distinguish between molecules of the same chemical composition.
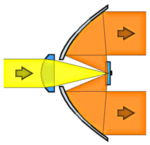
We also have to mention another important effect of the line-narrowing at low temperatures: The absorption cross section of the chromophores can become orders of magnitude larger than at room temperature since there is a better overlap between the narrow laser emission and a narrow molecular absorption line. (At room temperature the laser line only “covers” a very small part of the broad absorption spectrum of a typical chromophore.) Therefore one can often make do with simpler optics under cryogenic conditions because the interaction with the fluorophores is so much stronger and more selective. For an example we refer to the picture on the right hand side which shows the lens-paraboloid configuration with a simple singlet lens to focus the laser and a parabolic mirror to collect the fluorescence. The first ever fluorescence excitation experiment on a single molecule was performed in 1989 using such an apparatus. Another factor which explains why single molecules were first detected at liquid helium temperatures is the much increased stability of the single-molecule signals under these conditions.


